Lidocaine
In 1942 two Swedish chemists, Nils Löfgren and Bengt Lundqvist, discovered the compound we know today as “lidocaine.” It was clinically introduced by Dr. Torsten Gordh (1907-2010), who was the first physician in Sweden to specialize in anesthesiology. His clinical research with this new local anesthetic indicated that it was more effective and resulted in longer-lasting anesthesia when compared with other local anesthetics available at the time. He found a number of other advantages, including a lower risk of adverse effects, rapid onset, and stability when diluted and stored with epinephrine.
Astra AB, a small Swedish pharmaceutical company, acquired the rights to lidocaine in 1943 and gave the compound the brand name Xylocaine. The U.S. patent was granted in 1948 and Astra’s Worcester, Massachusetts manufacturing facility became operational in 1951. By this time Xylocaine was in very high demand. The popularity of this local anesthetic catalyzed Astra AB’s growth into a major international pharmaceutical company.
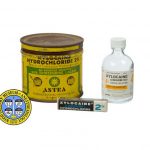
Over the years a number of formulations and packaging solutions have become available for the many types of anesthesia in which lidocaine has been put to use. Pictured here is a rusted tin which once contained 50 disposable cartridges of a lidocaine solution for injection. For use, the cartridges were placed into specially-designed reusable syringes. Also pictured is a box for a single glass ampule of an injectable solution, and a multidose bottle of a topical solution.
Catalog Record: Lidocaine
Access Key: akgk
Accession No.: 2001-03-04-1
Title: Xylocaine hydrochloride 2% (brand of lidocaine hydrochloride) / Astra
Pharmaceutical Products, Inc.
Corporate Author: Astra Pharmaceutical Products, Inc.
Title variation: Alt Title
Title: Lidocaine.
Publisher: Worcester, Mass. : Astra Pharmaceutical Products, [1951-1970].
Physical Descript: 1 tin : metal ; 8 x 9 cm. dia.
Subject: Anesthetics, Local.
Subject: Lidocaine.
Subject: Drug Packaging.
Note Type: General
Notes: Date range for the possible date of manufacture based on the year that the
Worcester, Massachusetts manufacturing plant was first operational and the
dates of publications where the word “tin” is still used to describe the
packaging for multiple cartridges of Xylocaine. The actual year of
manufacture should fall somewhere within this very generous date range. The
date range could change if reliable documentation indicates the range should
be corrected.
Note Type: Citation
Notes: Ball C, Westhorpe R. Local anaesthetics–The origins of lingnocaine. Anaesth
Intensive Care. 2004;32(5):611.
Note Type: Citation
Notes: Ball C, Westhorpe R. Local anaesthetics–The introduction of Xylocaine into
clinical practice.. Anaesth Intensive Care. 2004;32(6):733.
Note Type: Citation
Notes: Holmdahl MH. Xylocain (Lidocaine, Lignocaine), its discovery and Gordh’s
contribution to its clinical use. Acta Anaesthesiol Scand. 1998;42(suppl
113):8-12.
Note Type: Citation
Notes: Lindqvist K, Sundling S. Xylocaine – A Discovery – A Drama – An Industry.
[Södertälje]: Astra; 1993.
Note Type: Citation
Notes: Löfgren NM, Lundqvist BJ, inventors; Aktiebolaget Astra, Apotekarnes Kemiska
Fabrinker, assignee. Alkyl Glycinanilides. US patent 2,441,498. May 11, 1948.
Note Type: Citation
Notes: Wildsmith JAW. LidocaieL a more complex story thatn ‘simple’ chemistry
suggests. Proc Hist Anaesth Soc. October 9, 2010;43:9-16.
Note Type: Physical Description
Notes: 1 cylindrical tin, or metal container, intended for the packaging of 50
cartridges of local anesthetic; The metal of the tin is rusted; The tin is
approximately 7.6 cm in height and 8.7 cm in diameter; The outer
circumference of the tin is covered with product information that appears to
have been applied as paint or ink; The color of the label is yellow, with
green and black markings; Text on the primary area of the label includes, but
is not limited to the following, “XYLOCAINE [new line] HYDROCHLORIDE 2% [new
line] (Brand of Lidocaine Hydrochloride) [new line] AN AQUEOUS SOLUTION [new
line] with EPINEPHRINE 1:50,000 [new line] logo indicating the product was
accepted by the American Dental Association, the ASTRA logo and another logo
indicating the product was accepted by the American Dental Association [new
line] ASTRA [new line] PHARMACEUTICAL PRODUCTS, INC. [new line] Worcester,
Mass.; Markings to the right of the primary area on the label include, “1.8
cc min. U.S. Pat. No. 2,441,498 [new line] To be sold only as an unbroken
package [new line] Each cc contains: Xylocaine hydrochloride
(diethylaminoacet-2, 6-xylidide HCl) 0.02 Gm. [new line] Epinephrine 0.
00002 Gm. [new line] Sodium chloride 0.006 Gm. [new line] Sodium pyrosulfite
0.0005 Gm. [new line] Methylparaben 0.001 Gm. [new line] Water for
injection q. s. ad 1 cc [new line] Minimum contents of each cartridge 1.8 cc
[new line] MADE IN U.S.A.”; Markings to the left of the primary area on the
label include, “Caution: Federal Law Prohibits dispensing without
prescription. 50 Cartridges” [new line] VACUUM PACKED” [new line] IMPORTANT
[new line] Xylocaine Hydrochloride (Astra) two per cent is a patent
anesthetic agent. For routine use in dentistry adequate anesthesia may
ordinarily be provided with one-half the volume commonly suggested for a two
per cent solution of procaine. Recommended doses should be exceeded only when
conditions indicate the need for especially profound or longer lasting
anesthesia. For detailed dosage instructions, read enclosed pamphlet
carefully.”; Numbers embossed into the bottom of the tin: “L9349 [new line]
5107”.
Note Type: Reproduction
Notes: Photographed by Mr. Steve Donisch on January 14, 2013; This tin was
photographed with two other lidocaine packages; a multidose bottle of a
lidocaine solution for topical use, and a small box made for a single glass
ampule of a lidocaine and epinephrine solution also for injection.
Note Type: Acquisition
Notes: Donated to the WLM by George S. Bause, MD.
Note Type: Historical
Notes: In 1942 at Stockholm University’s Institute of Chemistry, after years of
systematic research by a number of scientists, two chemists, Nils Löfgren and
Bengt Lundqvist, discovered the compound that we know today as lidocaine.
Toxicology tests performed in 1943 at the Karolinska Institute by
physiologist Leonard Goldberg indicated that the new local anesthetic not
only posed a lower risk of adverse effects: It seemed to be more effective
and result in longer lasting anesthesia than other local anesthetics
available at the time. Lidocaine was first used in the medical-surgical
setting by the first physician to specialize in anesthesiology in Sweden, Dr.
Torsten Gordh (1907-2010). He conducted numerous tests using lidocaine for a
number of local and regional anesthesia techniques. His findings were similar
to those of dentists at the Royal School of Dentistry in Stockholm who tested
lidocaine in the dental setting under the direction of Hilding Bjorn
(1907-1995). Both parties reported very successful results. They verified
what Goldberg’s initial tests indicated, and highlighted a number of other
advantages, including lidocaine’s rapid effect, and its stability when
diluted and stored with epinephrine.
Note Type: Historical
Notes: Astra AB a small Swedish pharmaceutical company acquired the exclusive rights
to produce, manufacture and license lidocaine in 1943. They gave the compound
the brand name Xylocaine and moved into development mode, managing
complicated patent applications in Sweden and elsewhere, developing methods
of production, establishing supplies of the raw materials required for large
scale production, determining additional research needs, and planning for
marketing, distribution, and expansion beyond Sweden. This kind of
development was an enormous undertaking for a small company. Soon after the U
S. patent was granted in 1948, Astra AB’s U.S. subsidiary, Astra
Pharmaceutical Products, Inc., completed a small scale market trial in the
Boston area. Dentists were provided with sample cartridges of Xylocaine and
printed materials. The dentists reported very positive results. Word spread
far beyond Boston and Xylocaine quickly came into high demand. Xylocaine used
in the U.S. was manufactured in Sweden until 1951 when their Worcester,
Massachusetts manufacturing facility became operational. The popularity of
this local anesthetic catalyzed Astra AB’s growth into a major international
pharmaceutical company.
Note Type: Historical
Notes: Xylocaine was first made available in three different solution strengths,
with or without epinephrine, and packaged in disposable cartridges. Over the
years a number of formulations and packaging solutions have become available
for the many types of anesthesia in which lidocaine has been put to use.
Described here is a tin for 50 disposable cartridges of a lidocaine and
epinephrine solution for injection. For use, the cartridges were placed into
specially designed reusable syringes.
Note Type: Publication
Notes: Eriksson LI, Frostell C, Irestedt L, Lindahl S, Weitzberg E. In memoriam:
Torsten Gordh (1907-2010). Anesthesiology. 2010;113(6):1437-1438.
Note Type: Exhibition
Notes: Chosen for the WLM website (noted March 12, 2013).
Access Key: akgj
Accession No.: 2013-01-21-3
Title: Xylocaine (Lidocaine) hydrochloride 2% / Astra Pharmaceutical Products, Inc.
Corporate Author: Astra Pharmaceutical Products, Inc.
Title variation: Alt Title
Title: Xylocaine hydrochloride (Lidocaine) 2%.
Title variation: Alt Title
Title: Lidocaine.
Publisher: Worcester, Mass. : Astra Pharmaceutical Products, [1962-1998].
Physical Descript: 1 box : paperboard, paper : 1.5 x 6 x 1.5 cm.
Subject: Anesthetics, Local.
Subject: Lidocaine.
Subject: Anti-Arrhythmia Agents.
Subject: Drug Packaging.
Note Type: General
Notes: Range of years for possible year of manufacture based on copyright date on
product literature tucked into box and the year that Astra AB’s name changed
to AstraZenica.
Note Type: With
Notes: With glass ampule that measures approximately 5.8 cm x 1.2 cm diameter; Text
printed in red on ampule includes, “XYLOCAINE HCL 2% [new line] (lidocaine
brand) [new line] EPINEPHRINE 1:100,000 [new line] 2cc [new line] ASTRA [new
line] Worcester 6, Mass. U.S.A.”
Note Type: Citation
Notes: Ball C, Westhorpe R. Local anaesthetics–The origins of lingnocaine. Anaesth
Intensive Care. 2004;32(5):611.
Note Type: Citation
Notes: Ball C, Westhorpe R. Local anaesthetics–The introduction of Xylocaine into
clinical practice.. Anaesth Intensive Care. 2004;32(6):733.
Note Type: Citation
Notes: Gordh T, Gordh TE, Lindqvist K. Lidocaine: the origin of a modern local
anesthetic. Anesthesiology. 2010;113(6):1433-1437.
Note Type: Citation
Notes: Holmdahl MH. Xylocain (Lidocaine, Lignocaine), its discovery and Gordh’s
contribution to its clinical use. Acta Anaesthesiol Scand. 1998;42(suppl
113):8-12.
Note Type: Citation
Notes: Lindqvist K, Sundling S. Xylocaine – A Discovery – A Drama – An Industry.
[Södertälje]: Astra; 1993.
Note Type: Citation
Notes: Löfgren NM, Lundqvist BJ, inventors; Aktiebolaget Astra, Apotekarnes Kemiska
Fabrinker, assignee. Alkyl Glycinanilides. US patent 2,441,498. May 11, 1948.
Note Type: Citation
Notes: Wildsmith JAW. LidocaieL a more complex story that ‘simple’ chemistry
suggests. Proc Hist Anaesth Soc. October 9, 2010;43:9-16.
Note Type: Physical Description
Notes: 1 small paperboard box that opens on both ends; Blue, white and black print
or markings are on every side of the box; The ‘front’ is marked with,
“XYLOCAINE® [new line] (LIDOCAINE [new line] HYDROCHLORIDE”; To the right of
this text is marked, “2%”; Other markings on the box include an Astra logo on
each end of the box; Other text printed on the box includes, “Sterile Aqueous
Injection [new line] with EPINEPHRINE 1:100,000 [new line] Caution: Federal
law prohibits dispensing without prescription”, “Each cc. contains:
Diethylaminoacet-2, 6-xylidide HCL 0.02 Gm. [new line] Epinephrine 0.00001 Gm
[new line] Sodium chloride 0.006 Gm. [new line] Sodium metabisulfite 0.0005
Gm.
[new line] Sodium hydroxide to adjust pH”, “ASTRA PHARMACEUTICAL PRODUCTS,
INC. [new line] WORCESTER 6, MASS., U.S.A. [new line] U.S. PAT. NO. 2,441,498
PRINTED IN U.S.A. MADE IN U.S.A.”; A piece of paper, printed on both sides,
measuring approximately 9 x 5 cm in size is folded and tucked into the box
with the ampule; One side of the paper is printed with the same the
information that is printed on the outside of the box; The other side of the
paper is printed with the following text (for brevity, not all of the new
lines will be indicated), “LOCAL ANESTHETIC SOLUTION FOR INFILTRATIONS AND
MINOR NERVE BLOCK [new line] Usual Dose: 1-2 cc. [new line] Minor Surgical
Procedures: closure of minor lacerations, removal of small cysts, moles and
warts, treatment of abscesses. [new line] Therapeutic Nerve Blocks: as in
herpes zoster, subdeltoid bursitis, fibrositis, myalgia of shoulder muscles,
periarthritis due to trauma, painful postoperative scars. [new line]
Contraindications: Sepsis in the region of the proposed injection, severe
shock and heart block. Xylocaine (lidocaine) is usually employed in
combination with epinephrine to extend duration and to reduce systemic
absorption. In cases where vasopressors are clearly contraindicated, however,
only epinephrine free solutions should be used. [new line] Precautions:
Employ minimal effective dosage. INJECT SLOWLY, AND WITH FREQUENT ASPIRATION.
[new line] Untoward effects due to unusual sensitivity, faulty technique,
overdosage, and inadvertent intravascular or subarachnoid injection: Are
usually controlled by oxygen. Treat persistent convulsions, if and when seen,
with a short-acting barbiturate or a muscle relaxant. [new line] COPYRIGHT
1963 [new line] REVISED MAY, 1963”.
Note Type: Reproduction
Notes: Photographed by Mr. Steve Donisch on January 14, 2013; This bottle was
photographed with two other lidocaine packages; a tin for 50 disposable
cartridges of a lidocaine and epinephrine solution for injection, and a
multidose bottle for a lidocaine solution formulated for topical use.
Note Type: Historical
Notes: In 1942 at Stockholm University’s Institute of Chemistry, after years of
systematic research by a number of scientists, two chemists, Nils Löfgren and
Bengt Lundqvist, discovered the compound that we know today as lidocaine.
Toxicology tests performed in 1943 at the Karolinska Institute by
physiologist Leonard Goldberg indicated that the new local anesthetic not
only posed a lower risk of adverse effects: It seemed to be more effective
and result in longer lasting anesthesia than other local anesthetics
available at the time. Lidocaine was first used in the medical-surgical
setting by the first physician to specialize in anesthesiology in Sweden, Dr.
Torsten Gordh (1907-2010). He conducted numerous tests using lidocaine for a
number of local and regional anesthesia techniques. His findings were similar
to those of dentists at the Royal School of Dentistry in Stockholm who tested
lidocaine in the dental setting under the direction of Hilding Bjorn
(1907-1995). Both parties reported very successful results. They verified
what Goldberg’s initial tests indicated, and highlighted a number of other
advantages, including lidocaine’s rapid effect, and its stability when
diluted and stored with epinephrine.
Note Type: Historical
Notes: Astra AB a small Swedish pharmaceutical company acquired the exclusive rights
to produce, manufacture and license lidocaine in 1943. They gave the compound
the brand name Xylocaine and moved into development mode, managing
complicated patent applications in Sweden and elsewhere, developing methods
of production, establishing supplies of the raw materials required for large
scale production, determining additional research needs, and planning for
marketing, distribution, and expansion beyond Sweden. This kind of
development was an enormous undertaking for a small company. Soon after the U
S. patent was granted in 1948, Astra AB’s U.S. subsidiary, Astra
Pharmaceutical Products, Inc., completed a small scale market trial in the
Boston area. Dentists were provided with sample cartridges of Xylocaine and
printed materials. The dentists reported very positive results. Word spread
far beyond Boston and Xylocaine quickly came into high demand. Xylocaine used
in the U.S. was manufactured in Sweden until 1951 when their Worcester,
Massachusetts manufacturing facility became operational. The popularity of
this local anesthetic catalyzed Astra AB’s growth into a major international
pharmaceutical company.
Note Type: Historical
Notes: Xylocaine was first made available in three different solution strengths,
with or without epinephrine, and packaged in disposable cartridges. Over the
years a number of formulations and packaging solutions have become available
for the many types of anesthesia in which lidocaine has been put to use.
Described here is a small box made for a single glass ampule of a lidocaine
and epinephrine solution also for injection.
Note Type: Publication
Notes: Eriksson LI, Frostell C, Irestedt L, Lindahl S, Weitzberg E. In memoriam:
Torsten Gordh (1907-2010). Anesthesiology. 2010;113(6):1437-1438.
Note Type: Exhibition
Notes: Chosen for the WLM website (noted February 26, 2013).
Access Key: akgl
Accession No.: 2010-09-21-1 I
Title: Xylocaine 4% (lidocaine HCl) topical solution / Astra.
Corporate Author: Astra Pharmaceutical Products, Inc.
Title variation: Alt Title
Title: Xylocaine (lidocaine HCl) topical solution : 4%.
Title variation: Alt Title
Title: Lidocaine 4% topical solution.
Publisher: Worcester, MA : Astra Pharmaceutical Products, Inc., [1986-1988].
Physical Descript: 1 bottle : glass, plastic, paper ; 8.5 x 4.5 cm. dia.
Subject: Anesthetics, Local.
Subject: Lidocaine.
Subject: Drug Packaging.
Note Type: General
Notes: Range provided for possible year of manufacture is based on the expiration
date on the bottle: 3//1989. If the solution as packaged was presumed to have
a ‘shelf life’ of 3 years the date of manufacture would likely have been 1986
If the solution was presumed to have a shelf life of one year the year of
manufacture would likely have been 1988.
Note Type: Citation
Notes: Ball C, Westhorpe R. Local anaesthetics–The introduction of Xylocaine into
clinical practice.. Anaesth Intensive Care. 2004;32(6):733.
Note Type: Citation
Notes: Ball C, Westhorpe R. Local anaesthetics–The introduction of Xylocaine into
clinical practice.. Anaesth Intensive Care. 2004;32(6):733.
Note Type: Citation
Notes: Gordh T, Gordh TE, Lindqvist K. Lidocaine: the origin of a modern local
anesthetic. Anesthesiology. 2010;113(6):1433-1437.
Note Type: Citation
Notes: Holmdahl MH. Xylocain (Lidocaine, Lignocaine), its discovery and Gordh’s
contribution to its clinical use. Acta Anaesthesiol Scand. 1998;42(suppl
113):8-12.
Note Type: Citation
Notes: Lindqvist K, Sundling S. Xylocaine – A Discovery – A Drama – An Industry.
[Södertälje]: Astra; 1993.
Note Type: Citation
Notes: Löfgren NM, Lundqvist BJ, inventors; Aktiebolaget Astra, Apotekarnes Kemiska
Fabrinker, assignee. Alkyl Glycinanilides. US patent 2,441,498. May 11, 1948.
Note Type: Citation
Notes: Wildsmith JAW. LidocaieL a more complex story thatn ‘simple’ chemistry
suggests. Proc Hist Anaesth Soc. October 9, 2010;43:9-16.
Note Type: Physical Description
Notes: 1 glass bottle with a plastic screw-cap; It measures approximately 8.3 cm in
height and 4.2 cm diameter at the base; The plastic cap measures
approximately 2 cm in diameter; A white paper label with black and orange
markings is affixed to the bottle; Text on the primary area of the label
includes, “NDC 0186-0320-01 50 ml [new line] XYLOCAINE [new line] 4&
(LIDOCAINE HCl) [new line] TOPICAL SOLUTION [new line] NOT FOR INJECTION [new
line] Caution: Federal law prohibits [new line] dispensing without
prescription. [new line] ASTRA | Astra Pharmaceutical Products, Inc. [new
line] Worcester, MA 01606”; Text to the left of the primary area of the label
includes, “DO NOT INJECT [new line] 4% [new line] Consult package insert for
dosage and full prescribing information. [new line] Made and Printed in U.S.A
[new line] © Astra 1980 [new line] 070033-09 Rev. 11/81 (9)”; Text to the
right of the primary area of the label includes, “DO NOT INJECT [new line] 4%
[new line] Each ml contains: [new line] Lidocaine HCl 40 mg [new line]
Methylparaben 1 mg [new line] Sodium hydroxide to adjust pH [new line] Store
at controlled room temperature 15°-30°C (59°-86°F)”; Along the right edge of
the label is printed, “503046 [new line] EXP 3/89”. Embossed lettering on the
bottom of the bottle includes, “K [new line] T I [new line] 1 8”.
Note Type: Reproduction
Notes: Photographed by Mr. Steve Donisch on January 14, 2013; This bottle was
photographed with two other lidocaine packages; a tin for 50 disposable
cartridges of a lidocaine and epinephrine solution for injection, and a small
box made for a single glass ampule of a lidocaine and epinephrine solution
also for injection.
Note Type: Acquisition
Notes: Donated to the WLM by Hannsjoerg Hasche-Klunder, MD.
Note Type: Historical
Notes: In 1942 at Stockholm University’s Institute of Chemistry, after years of
systematic research by a number of scientists, two chemists, Nils Löfgren and
Bengt Lundqvist, discovered the compound that we know today as lidocaine.
Toxicology tests performed in 1943 at the Karolinska Institute by
physiologist Leonard Goldberg indicated that the new local anesthetic not
only posed a lower risk of adverse effects: It seemed to be more effective
and result in longer lasting anesthesia than other local anesthetics
available at the time. Lidocaine was first used in the medical-surgical
setting by the first physician to specialize in anesthesiology in Sweden, Dr.
Torsten Gordh (1907-2010). He conducted numerous tests using lidocaine for a
number of local and regional anesthesia techniques. His findings were similar
to those of dentists at the Royal School of Dentistry in Stockholm who tested
lidocaine in the dental setting under the direction of Hilding Bjorn
(1907-1995). Both parties reported very successful results. They verified
what Goldberg’s initial tests indicated, and highlighted a number of other
advantages, including lidocaine’s rapid effect, and its stability when
diluted and stored with epinephrine.
Note Type: Historical
Notes: Astra AB a small Swedish pharmaceutical company acquired the exclusive rights
to produce, manufacture and license lidocaine in 1943. They gave the compound
the brand name Xylocaine and moved into development mode, managing
complicated patent applications in Sweden and elsewhere, developing methods
of production, establishing supplies of the raw materials required for large
scale production, determining additional research needs, and planning for
marketing, distribution, and expansion beyond Sweden. This kind of
development was an enormous undertaking for a small company. Soon after the U
S. patent was granted in 1948, Astra AB’s U.S. subsidiary, Astra
Pharmaceutical Products, Inc., completed a small scale market trial in the
Boston area. Dentists were provided with sample cartridges of Xylocaine and
printed materials. The dentists reported very positive results. Word spread
far beyond Boston and Xylocaine quickly came into high demand. Xylocaine used
in the U.S. was manufactured in Sweden until 1951 when their Worcester,
Massachusetts manufacturing facility became operational. The popularity of
this local anesthetic catalyzed Astra AB’s growth into a major international
pharmaceutical company.
Note Type: Historical
Notes: Xylocaine was first made available in three different solution strengths,
with or without epinephrine, and packaged in disposable cartridges. Over the
years a number of formulations and packaging solutions have become available
for the many types of anesthesia in which lidocaine has been put to use.
Described here is a multidose bottle of a lidocaine solution for topical use.
Note Type: Publication
Notes: Eriksson LI, Frostell C, Irestedt L, Lindahl S, Weitzberg E. In memoriam:
Torsten Gordh (1907-2010). Anesthesiology. 2010;113(6):1437-1438.
Note Type: Exhibition
Notes: Chosen for the WLM website (noted February 26, 2013).